






























Phone: 626-365-0321
Email: atclean@atclean.com
Address:
9300 Santa Anita Ave, Ste 108
Rancho Cucamonga, CA 91730
USA
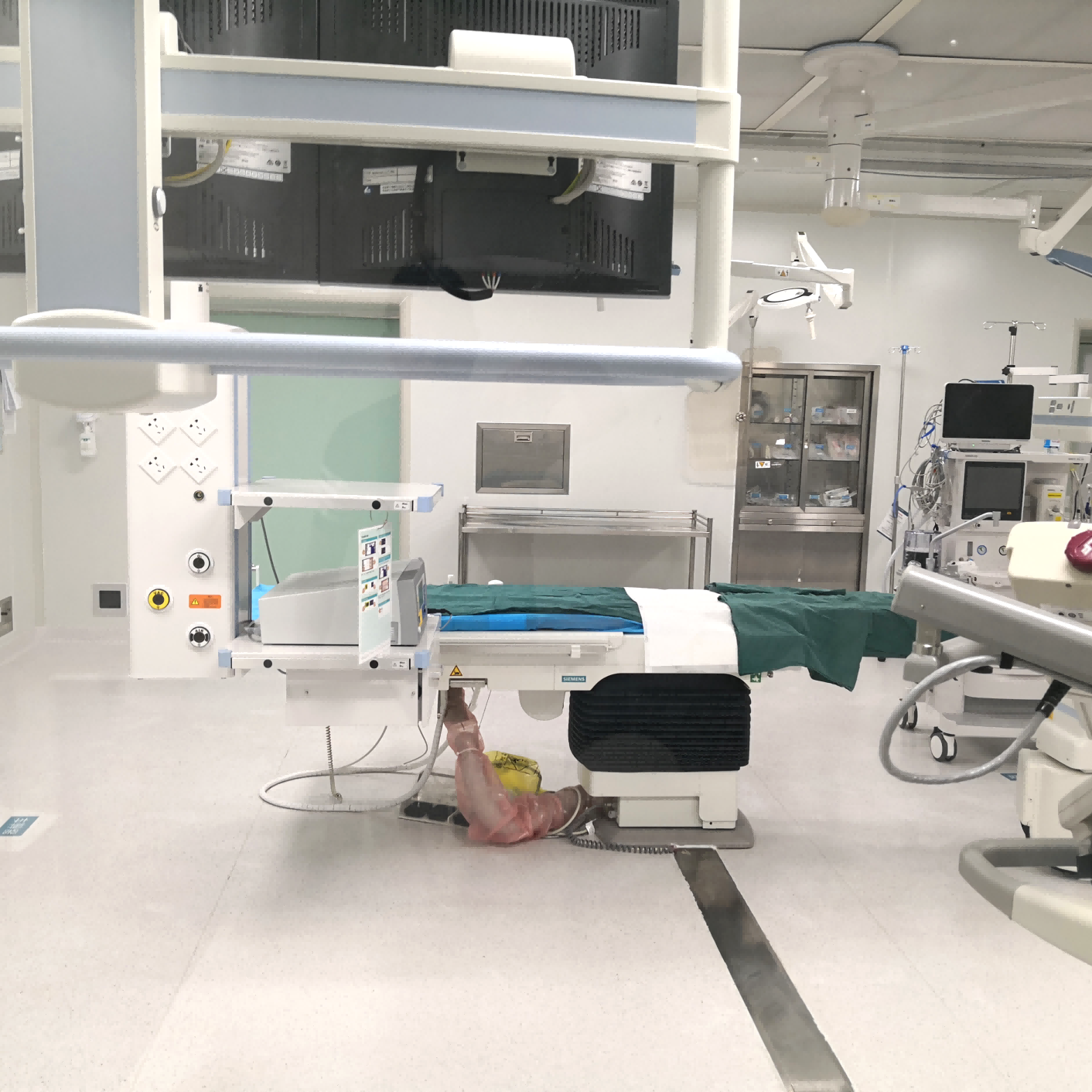
Silicon Valley Cleanroom Design Institute specializes in delivering cleanroom solutions for clients across semiconductor microelectronics, LED and LCD displays, optoelectronics, photovoltaic and chip manufacturing, precision manufacturing, food and pharmaceutical production, biological laboratories, hospital operating rooms, and aerospace applications.
Our team will contact you as soon as possible. For urgent assistance, please contact us at 909 266 3502.


